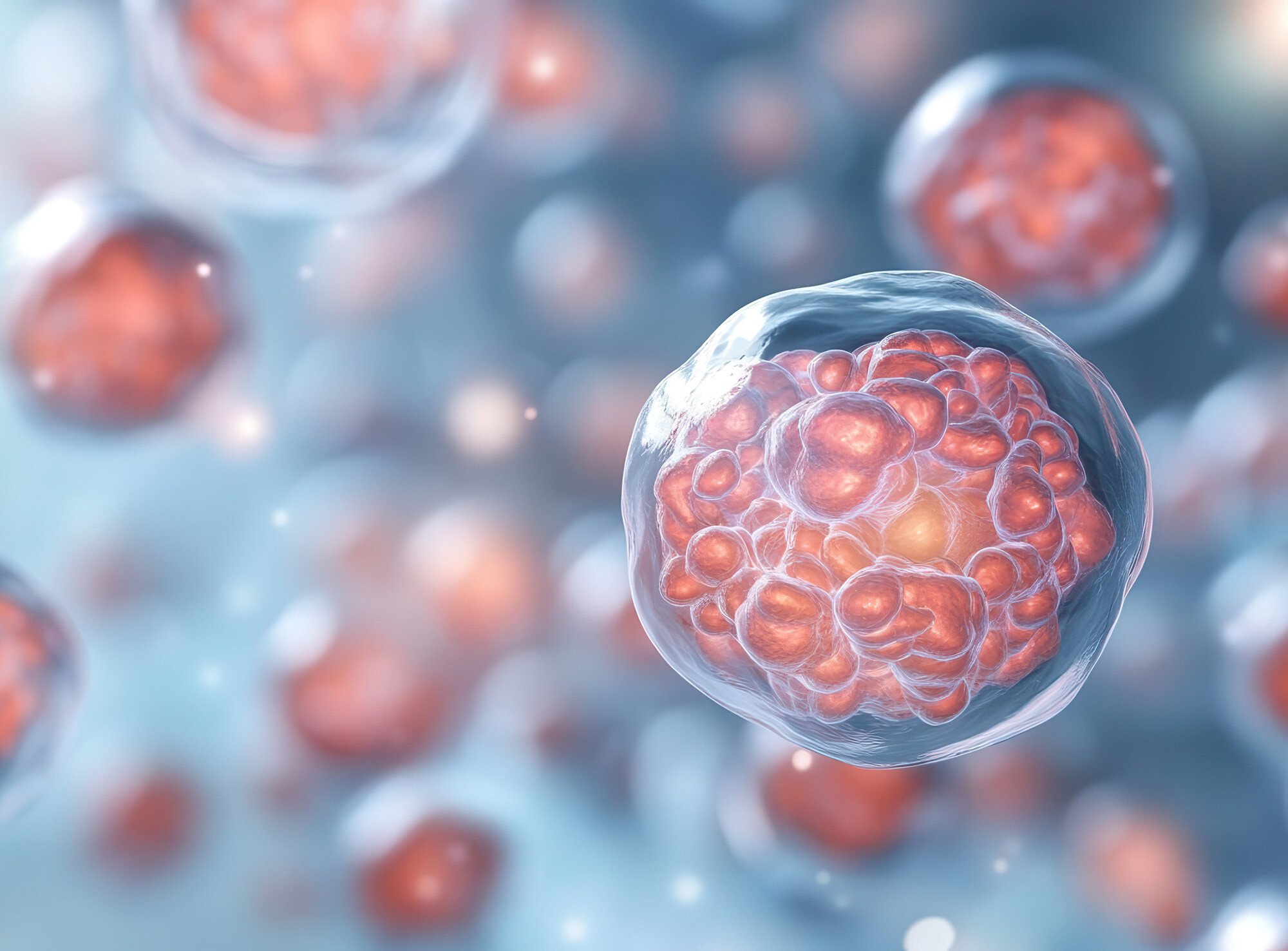
Grown from human pluripotent stem cells (iPSCs), organoids are three-dimensional (3D) cell clusters that form miniature organ-like structures. Because organoids mimic key functional, structural, and biological complexity of target organs, they hold great potential in organ development research, disease modeling, drug testing, and personalized medicine exploration.
The use of innovative 3D cell culture technologies is key to advancing organoid applications in regenerative medicine and clinical therapeutic development. Choosing the appropriate technology is important for effective culture expansion, maintenance of pluripotency and development of critical cellular attributes.
3D cell culture models can provide more control, lower manufacturing costs, and less labor-intensive operations compared to 2D culture systems. However, robust quality control systems are required to ensure that uniform aggregate size, shape, and growth rates are achieved as culture conditions adapt during large-scale expansion processes. Flow Imaging Microscopy (FIM) with FlowCam is a particle size and shape analyzer and is a highly efficient method for real-time high throughput assessment of 3D cell cluster attributes.
Download the Application Note: "Quality Control of Organoid and Tumoroid 3D Cell Clusters"
Advantages of Organoids and 3D Cell Cluster Technology
Two-dimensional (2D) cell cultures have been used in disease modeling for over 100 years; however, because of their inability to mimic the target tissue microenvironment, they often yield misleading results regarding cell behavior, drug responses, and disease progression1. The need for physiologically relevant human disease models is critical to bridging the gap between basic research and clinical applications and minimizing the risk of failed clinical trials.
iPSCs are rapidly addressing this need thanks to the increased efficiency and productivity of three-dimensional (3D) cell culture technologies2. With carefully controlled growth conditions, iPSCs can be reprogrammed to self-organize into miniature 3D organ-like structures, called organoids, that have the same complex cellular structures as the target tissue they are designed to mimic.
 Similarly, tumoroids, derived from patient tumor cells, can be grown in culture to maintain the full 3D architecture of the tumor microenvironment. The ability to model human tissues and tumors “in a dish” enables researchers to better understand how growth responses to culture conditions impact cellular differentiation mechanisms and viability.
Similarly, tumoroids, derived from patient tumor cells, can be grown in culture to maintain the full 3D architecture of the tumor microenvironment. The ability to model human tissues and tumors “in a dish” enables researchers to better understand how growth responses to culture conditions impact cellular differentiation mechanisms and viability.
The potential uses of organoids, tumoroids, and other 3D cell cluster culture systems in medicine present great promise in expediting drug discovery and advancing personalized medicine. And with the establishment of the FDA modernization act, 3D cell cluster technology is recognized as a safer, more efficient clinical trial option that is shifting disease model development away from the high costs and ethical pressures associated with whole animal testing.
Growing Organoids in Culture Presents Challenges
While the advantages of 3D cell culture in drug discovery, regenerative medicine, and disease modeling research have clear benefits, there are significant challenges in developing optimal culture conditions for preclinical model development and production scalability. Growth patterns of organoids and tumoroids can be highly variable, and the maturation of different cell types in culture can be difficult to control. Mimicking the complex microenvironment of the native tissue requires precise experimentation with growth factor cocktails and media formulations, scaffolding matrices, and culture technologies that support static versus dynamic culture conditions.
Depending on the type of 3D cell cluster desired, different growth requirements may be needed throughout the course of organoid growth. As the cells within the organoid mature, different signaling cues, growth factors, media components, and sometimes even the changes in the physical environment are required to effectively mimic the progression of development in vivo; an especially important requirement for long-term organoid cultures where significant changes in cell behavior can occur over time.
Innovative static and dynamic cell culture technologies that provide controlled microenvironments and improved nutrient delivery are being evaluated as versatile options for establishing appropriate microenvironments that improve culture growth and enable scalability. In static 3D cell cultures, cells are grown in a fixed, unchanging condition, typically within synthetic scaffolding matrices without any external forces or fluid flow. In dynamic 3D cell culture, culture conditions are actively manipulated to introduce fluid flow, mechanical stimulation, or other dynamic factors to mimic physiological conditions. These complex processes require effective monitoring of 3D cell cluster size, shape, and morphology metrics to ensure conditions are appropriately tuned and the desired developmental cell cluster fate is achieved.
Monitoring Organoid Growth with Flow Imaging Microscopy
Traditional microscopic analysis of 3D cell clusters is time-consuming and limited because it relies on qualitative visual inspection followed by digital analysis of only a handful of individual cell clusters. This method requires specialized expertise and, because of the limited sample size, fails to capture the full diversity of 3D aggregate morphological attributes.
Alternatively, utilizing advanced imaging technologies to monitor 3D cell cluster morphology and cellular behavior in real-time can provide valuable insights into optimization and quality control. Flow Imaging Microscopy, for example, offers an automated, high-throughput method for assessing organoid morphology, analyzing cellular heterogeneity, and informing overall system health. With FlowCam, the biological quality of organoids can be immediately determined, and efficient cell culture process strategy decisions can be made quickly and reliably.
 In our new application note, "Quality Control of Organoid and Tumoroid 3D Cell Clusters with FlowCam", we describe the advantages of using FlowCam to assess iPSC culture optimization methods and we present a study performed in collaboration with Corning Life Sciences. The study compares static 3D culture data obtained from traditional manual microscopy with data obtained via FlowCam. The ability to process many more particles and perform high-throughput data analysis in real-time with FlowCam provided an objective, more complete assessment of the aggregate population in a fraction of the time required by manual microscopy.
In our new application note, "Quality Control of Organoid and Tumoroid 3D Cell Clusters with FlowCam", we describe the advantages of using FlowCam to assess iPSC culture optimization methods and we present a study performed in collaboration with Corning Life Sciences. The study compares static 3D culture data obtained from traditional manual microscopy with data obtained via FlowCam. The ability to process many more particles and perform high-throughput data analysis in real-time with FlowCam provided an objective, more complete assessment of the aggregate population in a fraction of the time required by manual microscopy.
References:
- Ferreira LP, Gaspar VM, Mano JF. Design of spherically structured 3D in vitro tumor models—Advances and prospects. Acta Biomater. 2018 Jul 15;75:11-34. https://doi.org/10.1016/j.actbio.2018.05.034
- Kim J, Koo BK, Knoblich JA. Human organoids: model systems for human biology and medicine. Nat Rev Mol Cell Biol. 2020;21:571–584. https://doi.org/10.1038/s41580-020-0259-3