
Effective strategies for monitoring particles in biotherapeutics are critical to meet regulatory requirements like USP <788> and mitigate the …
Read Post

A central challenge with particle analysis in biotherapeutics is the wide size range of particles that may be present. Particles in these samples can …
Read Post

Monitoring particles in biotherapeutics during manufacturing is critical to ensure product quality and to meet regulatory requirements on particle …
Read Post

Particles are ubiquitous in biotherapeutics and can drastically impact the efficacy of a given drug. Therefore, characterizing them is a critical …
Read Post
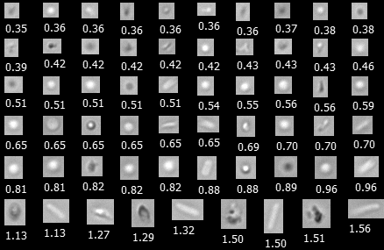
Many new types of biotherapeutic Active Pharmaceutical Ingredients (API) such as viruses, nanomedicines including virus-like particles and lipid …
Read Post
Silicone oil has long been used in the pharmaceutical industry as a lubricant for syringes. It serves to reduce the force necessary to depress the …
Read Post

Pollen-based microcapsules such as hollow sporopollenin exine capsules (SECs) have emerged as excellent drug delivery and microencapsulation vehicles …
Read Post
Injectable biopharmaceuticals, particularly prefilled syringes (PFS), come with risks related to the force of injection, such as patient pain and …
Read Post

A recent study by researchers from the University of New England and University of New Hampshire has demonstrated that flow imaging microscopy is an …
Read Post






